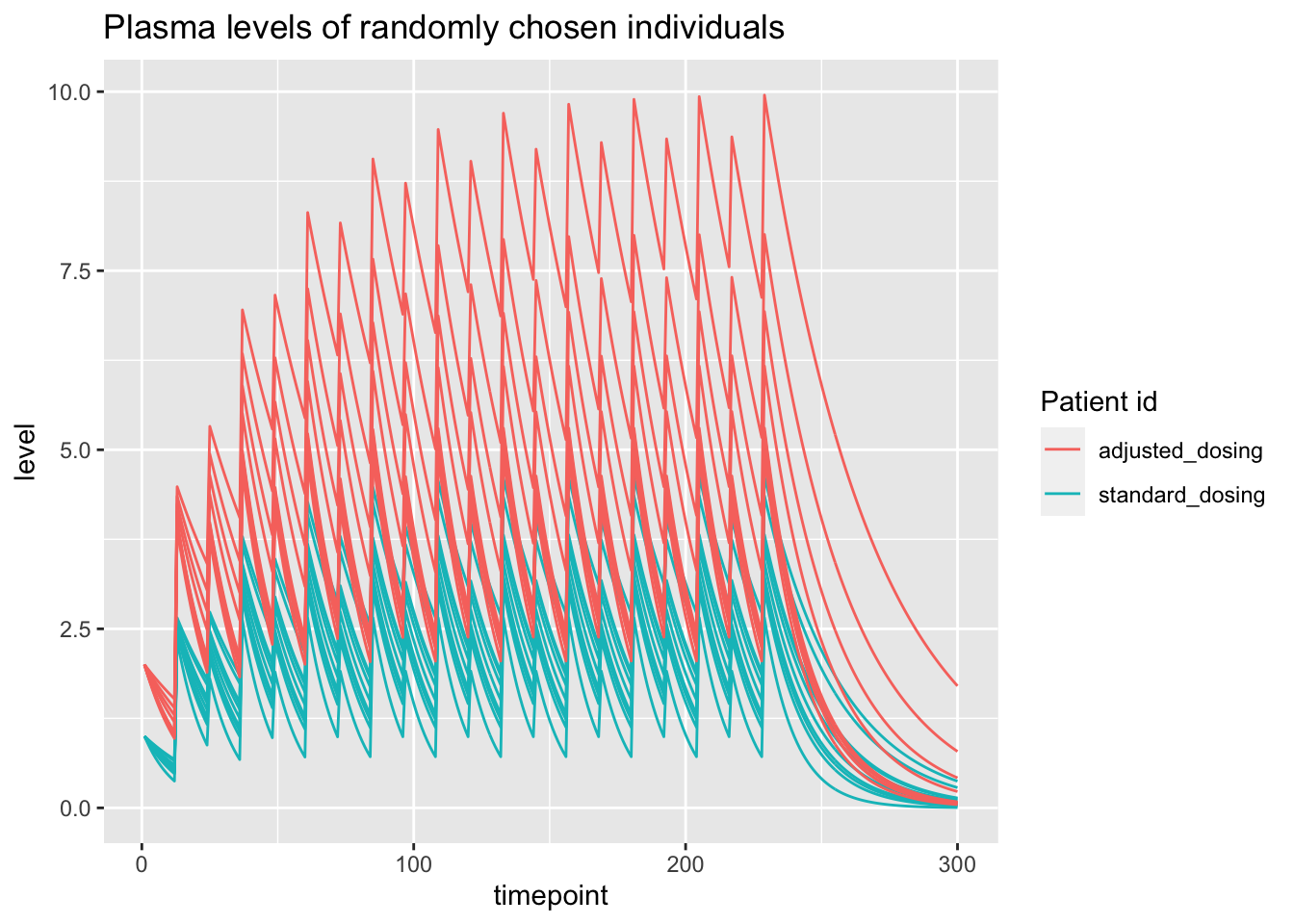
set.seed(123)
nr_of_patients <- 200
patients <- tibble(id = 1:nr_of_patients,
group = rep(c('standard_dosing', 'adjusted_dosing'), each = nr_of_patients / 2),
age = rnorm(nr_of_patients, mean = 60, sd = 10),
weight = rnorm(nr_of_patients, mean = 80, sd = 10),
age_scaled = scale(age)[, 1],
weight_scaled = scale(weight)[, 1],
sex = sample(c('male', 'female'),
size = nr_of_patients,
replace = TRUE),
leukemic_burden_pretherapy = rlnorm(nr_of_patients))
patients |>
sample_n(5) |>
kable(align = 'c')| id | group | age | weight | age_scaled | weight_scaled | sex | leukemic_burden_pretherapy |
|---|---|---|---|---|---|---|---|
| 182 | adjusted_dosing | 72.63185 | 81.90230 | 1.3483988 | 0.1487158 | female | 2.670092 |
| 13 | standard_dosing | 64.00771 | 92.32476 | 0.4340111 | 1.1952301 | male | 2.416850 |
| 68 | standard_dosing | 60.53004 | 62.43473 | 0.0652855 | -1.8060152 | female | 1.798109 |
| 127 | adjusted_dosing | 62.35387 | 79.39178 | 0.2586592 | -0.1033647 | female | 1.186002 |
| 25 | standard_dosing | 53.74961 | 76.36343 | -0.6536207 | -0.4074403 | male | 3.048497 |